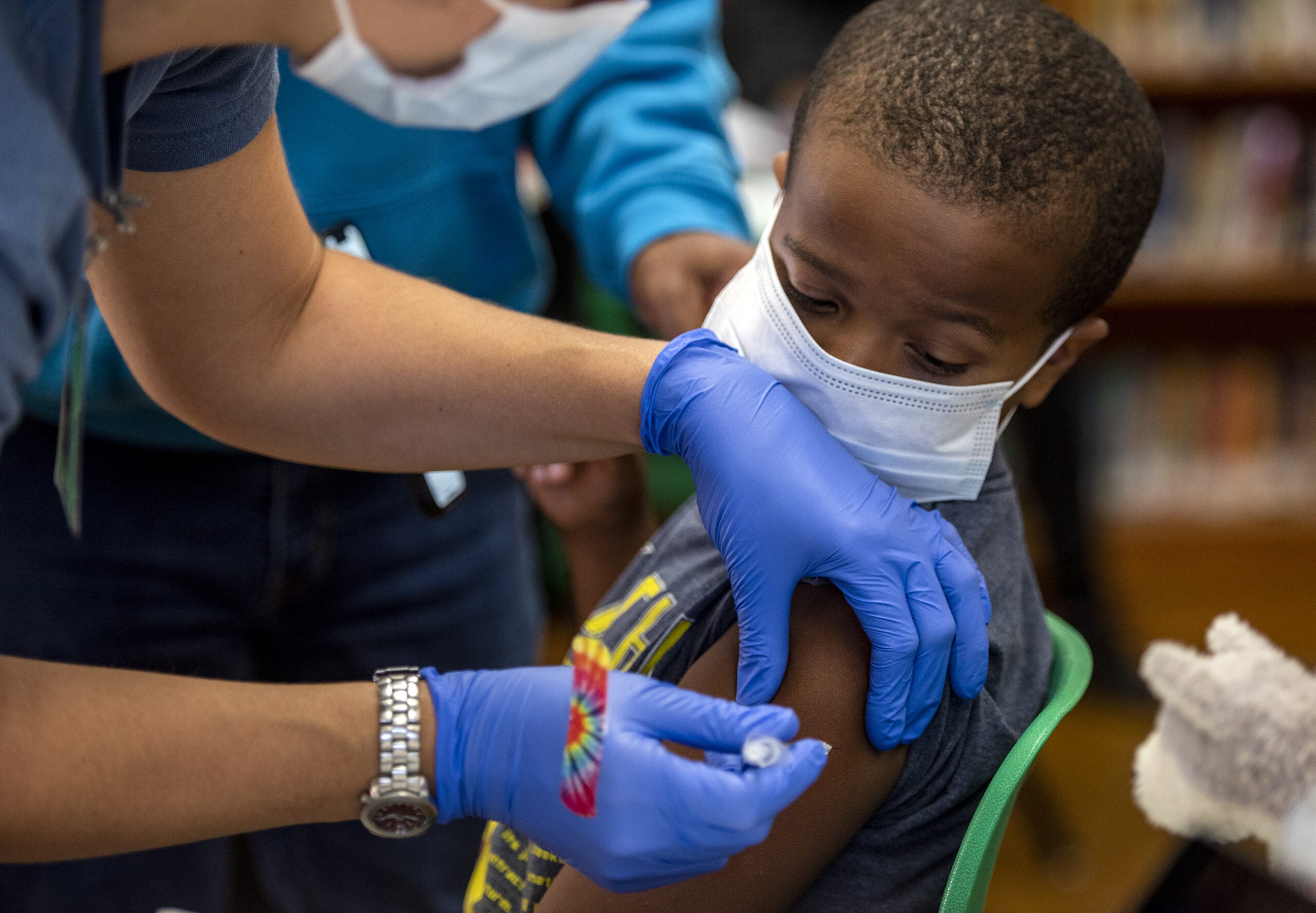
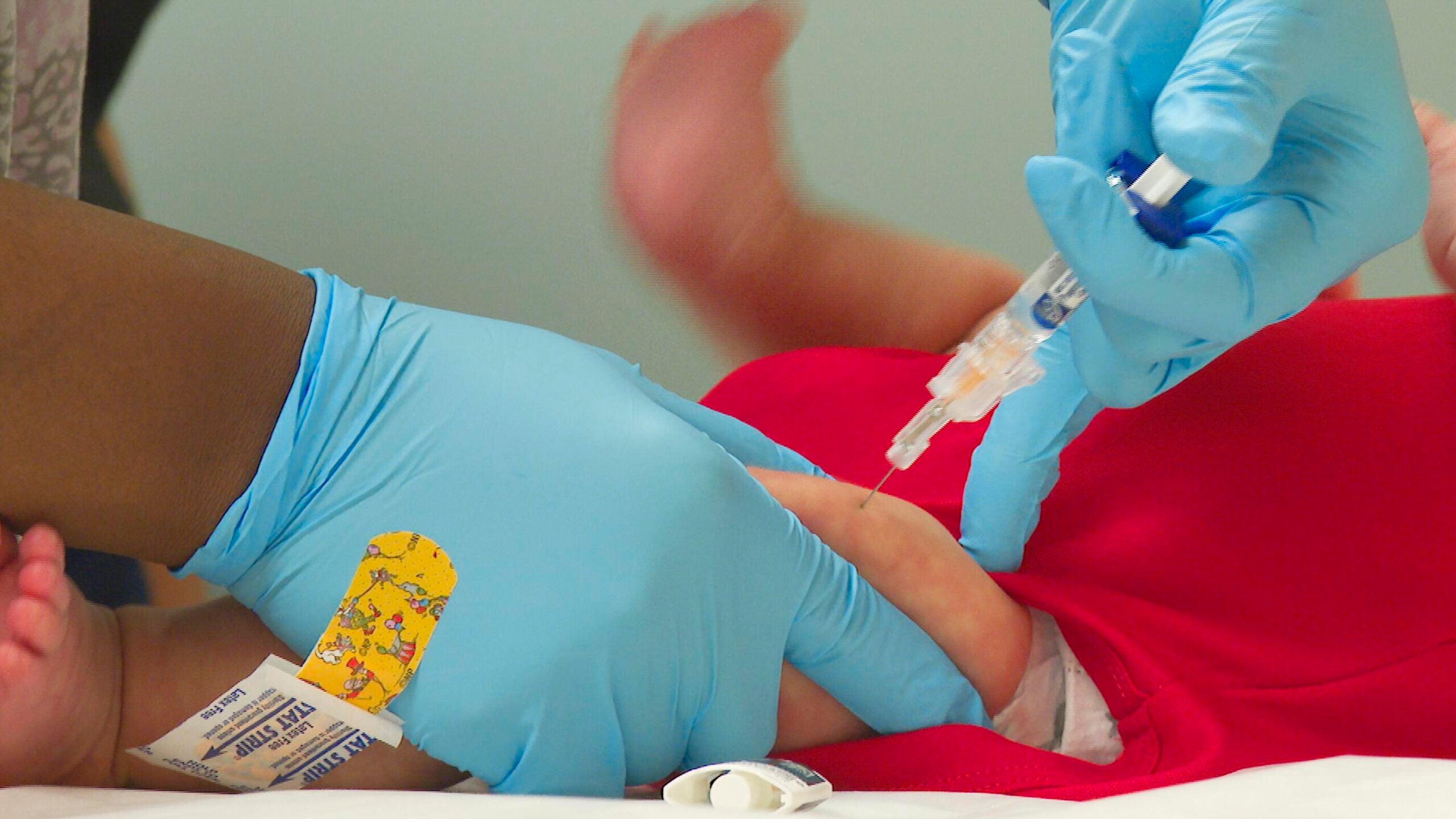
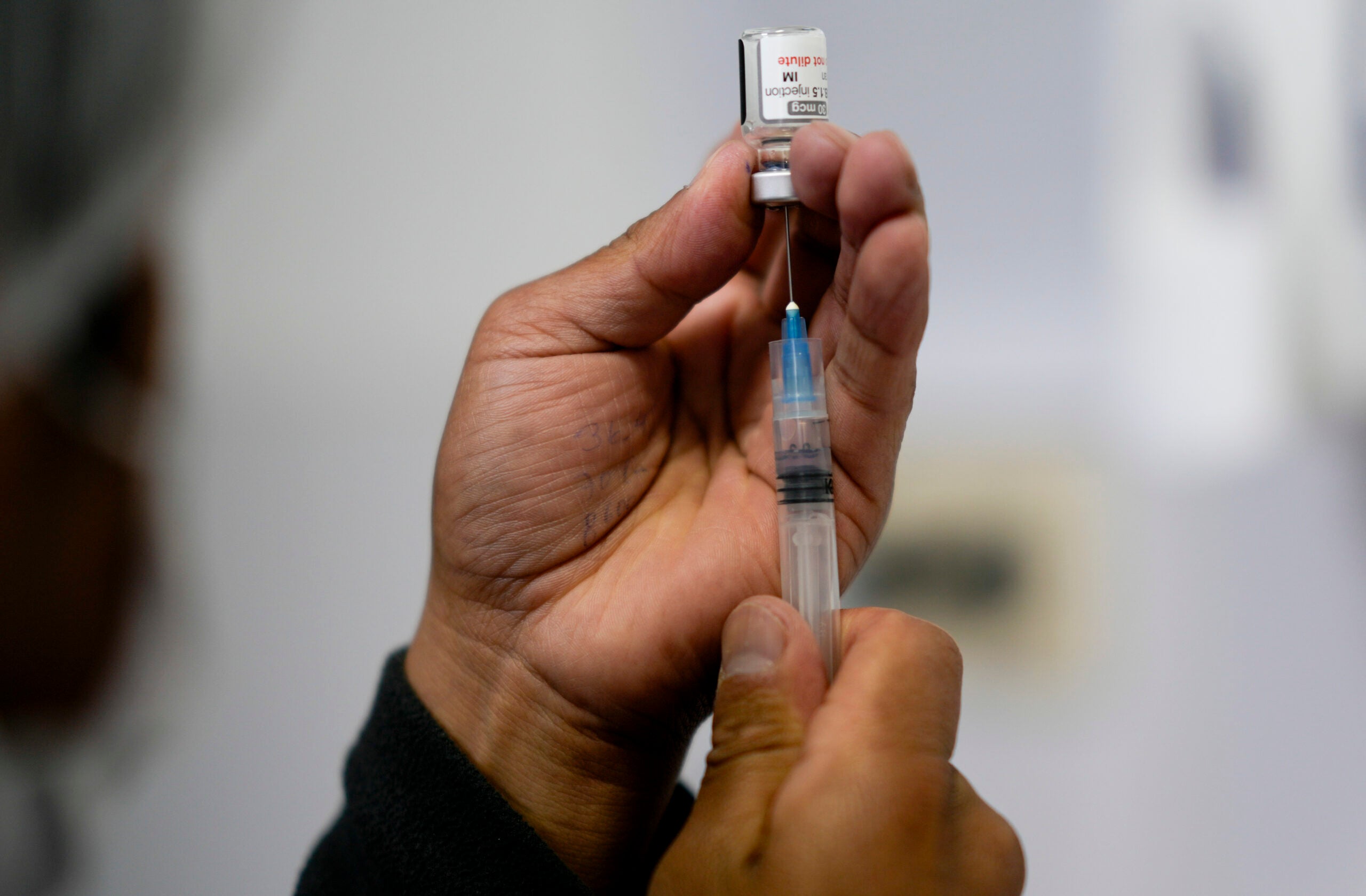
The COVID-19 manufacturer Moderna said Wednesday that it plans to submit a request to the Food and Drug Administration to authorize its vaccine for kids ages 6 and under “in the coming weeks.”
It’s a promising sign for Wisconsin families who have been waiting on a vaccine for their young children, though many are wary of getting their hopes up.
At the end of February, Pfizer released early findings on its vaccine for kids under 5 that showed protection from the shot declined quickly in kids, pushing back the date kids might be able to get their shots.
News with a little more humanity
WPR’s “Wisconsin Today” newsletter keeps you connected to the state you love without feeling overwhelmed. No paywall. No agenda. No corporate filter.
That made Appleton mom Annette Pieper reluctant to get too excited about the Moderna vaccine being available to her 4-year-old son Oliver soon.
“It’s been frustrating, because you hear it’s going to be soon, before the end of the year, and then it’s pushed back — and then it’s going to be early in the year, and then it’s pushed back,” she said. “I’m very cautiously optimistic, but I’m not excited yet.”
Pieper and her family have been masking up in grocery stores and on the school bus, which sometimes leads to dirty looks or comments, she told WPR’s WHYsconsin. She said they’ll keep taking precautions after Oliver gets his shots, but they’ll feel better about his safety and that of people around him.
“I’m not expecting it to be, we can just throw our masks away forever, and run around and have big parties,” she said. “But I’ll be able to sleep a little easier at night knowing he has a good solid base level of protection.”
The pediatric vaccine that Moderna plans to submit for approval is one-quarter the size of the first dose adults receive. It will also be a two-shot sequence, and will be available for kids between 6 months and 6 years old. No severe cases of COVID-19 were reported among the vaccinated children in Moderna’s trials.
Pieper said she’d like to take Oliver to see his paternal grandparents, who live near Chicago, but since they’d want to spend a couple nights, they’ve been putting it off until he’s vaccinated. They’ve been able to bring Oliver to see some family in Menasha and Ripon, but only for short periods.
“When I see them, I’m always thinking, ‘OK, is anyone sick, are we gonna get anyone sick?’” she said. “We’re still trying to be very careful.”
On Wednesday, the same day Moderna announced it would submit its shot for FDA approval, Pieper was also breaking out one of her at-home tests because she heard from Oliver’s school that he had a cough and would need to be cleared before he could go back to school.
“The timing of it,” she said, laughing ruefully.